Calculate the theoretical percentage of water in gypsum, rounded to the nearest whole number %. (calcium sulfate dihydrate; CaSO4 ° 2H2O; Ca = 40 g/mol, .... 2H2O. (1) What is the name given to the water molecules present in the salt? (ii) Calculate the percentage of water molecules in hydrated calcium sulphate .... Sep 17, 2012 — Gypsum has the formual CaSO4 2H2O. What is the percent by weight of water in this hydrate? If 15.000 grams of this hydrate is heated, what ...
Question: Calculate theoretical percentage by mass of water in the hydrate CaSO4•2H2O. Show your work. This problem has been solved! See the answer .... stated: the molar mass of water is 18.02 g/mol. Notice that ... Using Molar Mass calculations to figure the percentage that ... Percent water in CaSO4•3H2O.
Jada, 735C5E05-C080-42F9-9C24-5B8EAC90 @iMGSRC.RU
Calculate the theoretical percentage of water in gypsum (calcium sulfate dihydrate, CaSOs 2H20) 2. A student reported the following values for the percentage of .... Jan 23, 2020 — The percent composition of water in the given sample is 20.93 %. Explanation: To calculate the percentage composition of chlorine in sample, .... Sep 19, 2017 — In the laboratory, the hydrate CaSO4 2H2O(s) can be heated in a crucible to completely drive off the water of hydration to form the anhydrous ... a-youtube-downloader-free-xpe-x-ray
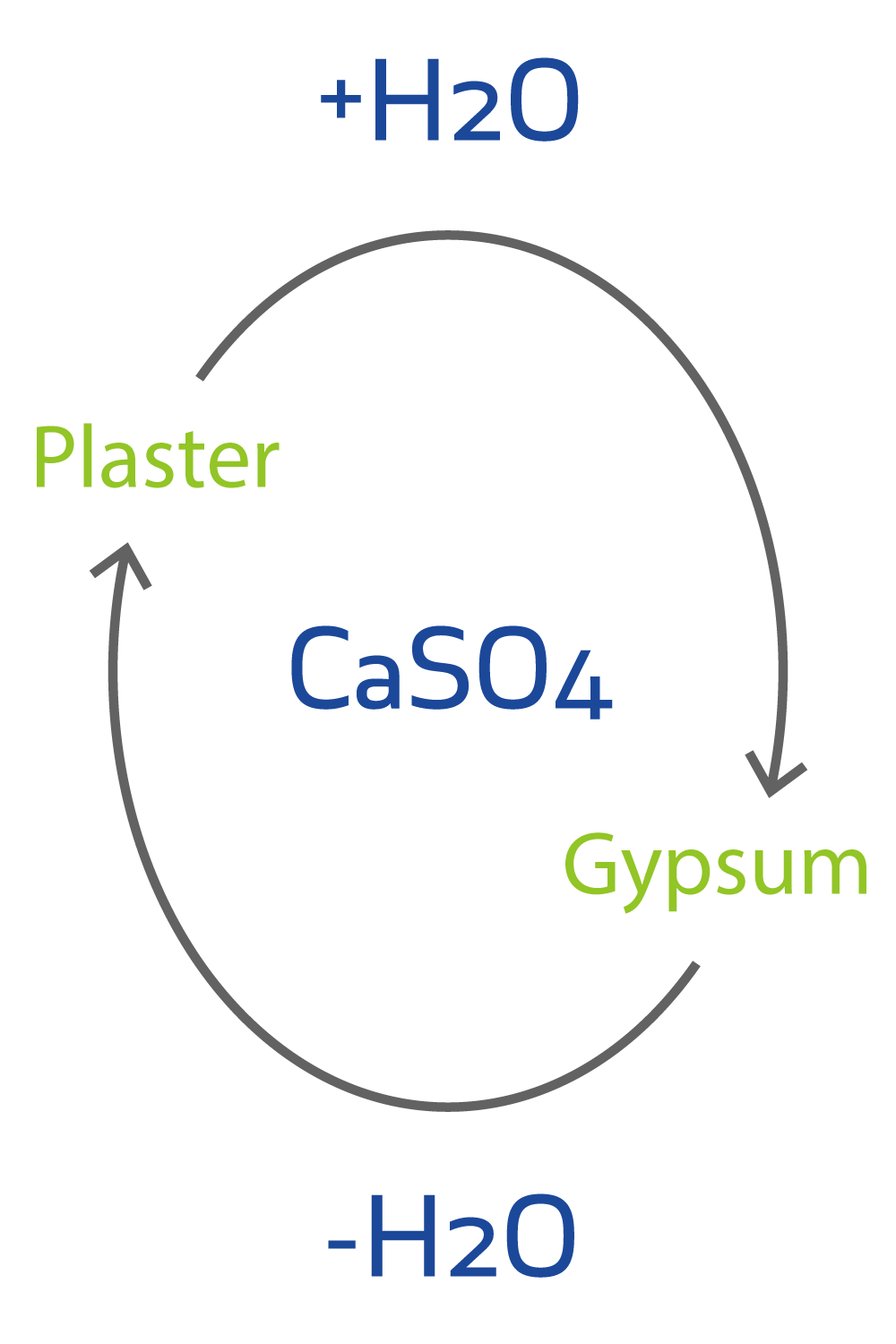
Percent Composition o Use to verify purity of a sample; mass percent of each element in a compound: ... Determine moles of anhydrate (from formula and molar mass) and moles of water ... Formula = CaSO4 · 2H2O: calcium sulfate dihydrate.. Convert between CaSO4*2H2O weight and moles ... Elemental composition of CaSO4*2H2O ... Mass percent composition, Atomic percent composition. Ca S O .... ... need a definite formula before I can calculate its percentage composition, i.e. its hydration . ... %water=mass of waterMass of calcium sulfate dihydrate×100%=.. Hydrates are substances formed when water combines chemically in definite proportions ... CaSO4 • 2H2O ... (g) Percentage of water in hydrate [(f) ÷ (c)] × 100. CRACK RHINOCEROS V6.5.18149.14421 CRACK [Crackzsoft]
(calcium sulfate dihydrate; CaSO4 ° 2H2O; Ca = 40 g/mol, S= 32 g/mol; O= 16 g/mol; H= 1 g/mol).;;. tina. Feb 6, 2013. %H2O = (mass H2O/mass .... Calculate the molar mass of CaSO4.2H2O in grams per mole or search for a chemical formula or ... Element, Symbol, Atomic Mass, # of Atoms, Mass Percent.. Gypsum is calcium sulphate dihydrate CaSO4.2H2O Calculate the molecular weight Ca=40.078 S=32.06 ... therefore the percentage of water in gypsum is. Results for: CaSO4.2H2O. Element, Number, Mass, Percet ... Total MW = 138.1658, Total percent composition = 100. Would you like to enter another formula?. Answer to: What is the mass percent of water in plaster of Paris, CaSO4 1/2H2O? By signing up, you'll get thousands of step-by-step solutions to.... Plaster of Paris (Ca(SO4).1/2H2O). More... Molecular Weight. 290.3. Component Compounds. CID 5460341 (Calcium). CID 1118 (Sulfuric acid). CID 962 (Water).. The chemical formula for gypsum is CaSO4 • 2H2O and the chemical name is calcium sulfate dihydrate. Note that the dot in the formula (or multiplication sign) .... To find the percent composition (by mass) for each element in CaSO4 . 2H2O we need to determine the ... 3e88dbd8be Craftsman electric staple gun manual

